Stimuli Technology - Solution de traitement des patients atteints d’hyperactivité de la vessie
Sensibiliser la population aux symptômes d'hyperactivité vésicale
Sa mission est d’améliorer la vie des patients atteints d’hyperactivité vésicale grâce à une technologie basée sur la neurostimulation électrique transcutanée, plus communément appelée TENS (Transcutaneous Electrical Nerve Stimulation).
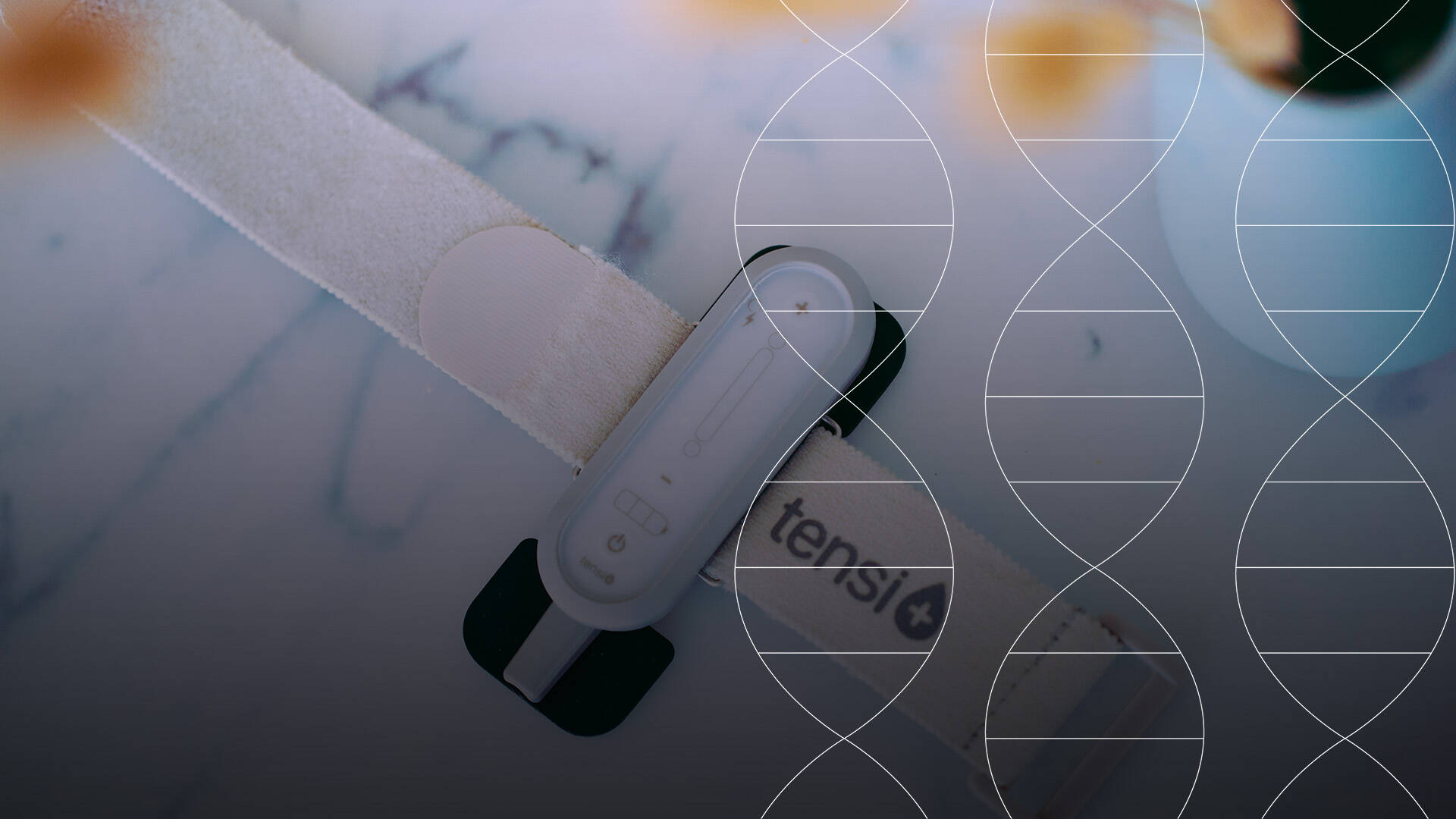
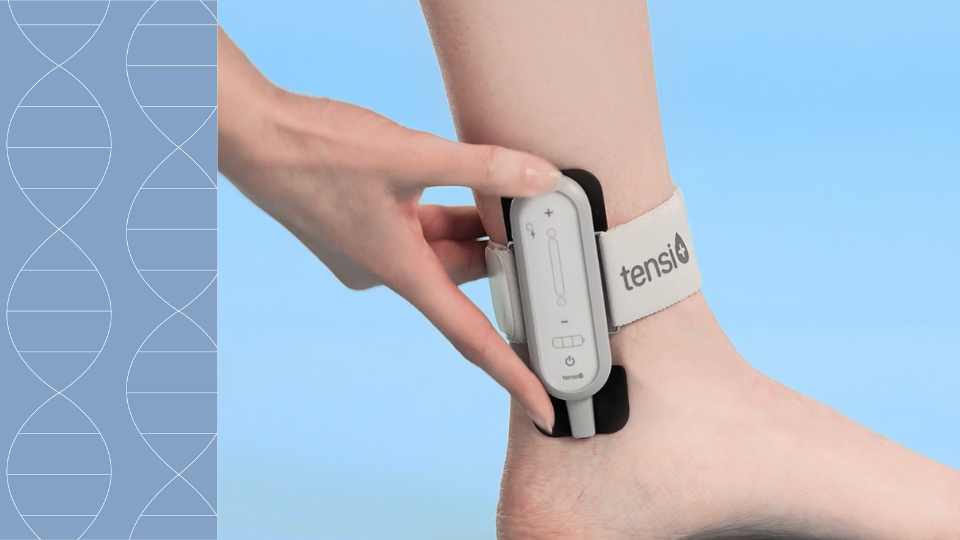
Pour répondre à ce besoin, Stimuli Technology a développé Tensi+, un dispositif médical traitant l’hyperactivité vésicale.
Ce traitement non invasif placé sur le passage du nerf tibial postérieur envoie des stimulations électriques par l’intermédiaire d’électrodes situées sur la peau.

Ambition
Améliorer la vie des patients atteints d’hyperactivité vésicale
L’hyperactivité vésicale touche aujourd’hui aussi bien les hommes que les femmes et peut survenir à tout âge. Il se manifeste principalement par l’envie fréquente, soudaine et incontrôlable d’uriner ; ces symptômes pouvant survenir à n’importe quel moment du jour ou de la nuit.
Les études ont montré que 15%* de la population est atteinte par l’hyperactivité vésicale, aujourd’hui sous-diagnostiquée.
Désormais bien implantée en France et reconnue par plusieurs médecins urologues de renom, l’équipe Stimuli Technology s’attaque désormais au marché européen.

L’équipe dirigeante de Stimuli Technology
Témoignages
Envie de rejoindre l’équipe de Stimuli Technology ?
Kit de presse
Vous êtes journaliste ? Téléchargez le dossier de presse de Stimuli-Technology
Stimuli Technology - Dossier de presse Septembre 2022Découvrez les actualités de Stimuli Technology
Santé
Améliorer le quotidien des praticiens et l'efficacité des soins
Déployer de nouvelles solutions technologiques et industrielles auprès des professionnels de santé afin d’améliorer le bien-être des patients et le quotidien des praticiens.